| Monthly Tech-Tip | No tracking! No ads! No paywall! |
Gerstley Borate vs Frit 3134 melt fluidity comparison
This GLFL test compares the melting behavior of Gerstley Borate (GB) and Ferro Frit 3134 across a wide temperature range. Although both supply boron, they behave very differently because Gerstley Borate is a natural mineral blend while Frit 3134 is a premelted glass. Gerstley Borate shrinks dramatically to about half its original size by 1600°F, then suddenly becomes highly fluid between 1600–1650°F as reactive borate phases form and viscosity collapses. Frit 3134, by contrast, softens gradually, reflecting the progressive viscosity decline typical of a glass. It has no crystal decomposition or raw material phase changes, no sudden liberation of fluxes. It is softening while gases are still escaping (the GB was also foaming at 1650F).
So, if GB delivers boron through mineral breakdown and 3134 delivers it through viscosity-controlled glass softening, then why does a zero LOI frit have bubbles at 1700F? The dried ball is a compacted powder; it has plenty of voids. This melt begins extremely early, at 1350F, easily trapping air in the voids and gases generated by the binder holding the frit ball together. While bubbles have cleared this fluid melt by 1800F, a bubble-free glaze is much more challenging because there are more sources of decomposition gases and a stiffer, more viscous melt.
Related Pictures
Gerstley Borate is passing on to a better place. With a 300% price hike.

This picture has its own page with more detail, click here to see it.
Gerstley Borate has just become Costly Borate. The supplier, LagunaClay.com, likely raised the pice to wake us all up to take action in substituting it before supplies run out. It is a ceramic glaze flux, sourcing boron to melt far better than any other common raw material. It has been a foundation material in low and middle temperature pottery glaze recipes for many decades. Potters have a love/hate relationship with it: Enjoying its low melting point but enduring its problems (inconsistency, gelling of the slurry, crawling, micro-bubbles, boron-blue discoloration). Strangely most people have used it without knowing what it really was. And few realize how easy it is to replace. Yes, existing substitutes work sometimes - but it is better to adjust each glaze recipe to source boron from a frit (fixing other issues also). Please read that last statement carefully. It did not say that there is any frit that can substitute. It said that frits can source boron.
Ferro Frit 3134 is NOT A SUBSTITUTE for Gerstley Borate

This picture has its own page with more detail, click here to see it.
This frit, or any of similar chemistry (e.g. Fusion F-12) IS NOT A 1:1 SUBSTITUTE for Gerstley Borate, their chemistries are too different. That being said, the frit sources lots of B2O3, that makes it a candidate to weave into recipes as the source of boron. To show the difference I have put 100 parts of each in side-by-side recipes in my Insight-live.com account, set the calculation type to non-unity formula and increased the frit until the B2O3 in the two match. Notice it takes 118g of the frit to source the same amount of B2O3 as 100 GB. Notice also that the frit sources almost triple the amount of Na2O per weight unit (that is a big deal because it means the recipe containing the Gerstley Borate to be substituted needs an Na2O-sourcing material that can be reduced to compensate). And the frit sources 3.5 times the SiO2 (other SiO2-sourcing materials in the recipe will need to be cut to compensate). And the Gerstley Borate has significant MgO while the frit has none (so an MgO-sourcing material like talc will be needed). Minor tweaks will also be needed to reduce other sources of CaO (since the frit has quite a bit more). The recipe will also need enough flexibility to do the final matching of Al2O3 and SiO2. The GBMF test confirms the difference at 1700F, these 10-gram balls melted down onto the tiles very differently.
Why does Gerstley Borate melt in two stages? Because it is two minerals.
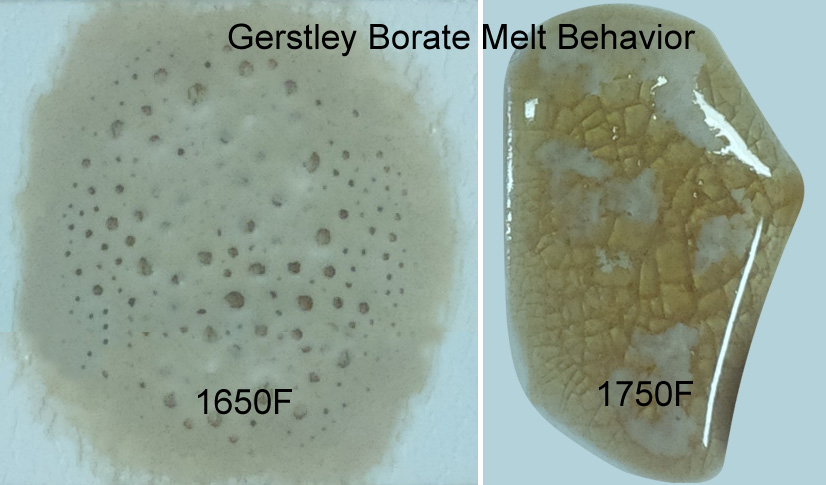
This picture has its own page with more detail, click here to see it.
The ulexite in Gerstley Borate melts first, producing an opaque fired glass having the unmelted (and still gassing) particles of colemanite suspended in it. By 1750F the colemanite is almost melted also. Boron-containing frits, by contrast, soften slowly over a wide temperature range and gradually spread and melt. Not surprisingly they produce a more stable glaze (albeit often less interesting visually without additives e.g. titanium, rutile). These behavior contributes to phase changes in fired glazes that contribute to variegation.
Can you actually throw a Gerstley Borate glaze? Yes!

This picture has its own page with more detail, click here to see it.
G2931 Worthington Clear is a popular low to medium-fire transparent glaze recipe. It contains 55% Gerstley Borate (GB) plus 30% kaolin (GB melts at a very low temperature). GB is also very plastic, like a clay. I have thrown a pot from this glaze recipe! This explains why high Gerstley Borate glazes often dry so slowly and shrink and crack during drying. When recipes also contain a plastic clay like this one the shrinkage is even worse. GB is also slightly soluble, over time it gels glaze slurries even in smaller percentages. Countless potters struggle with Gerstley Borate recipes.
Why so many bubbles in a fritted cone 6 glaze?

This picture has its own page with more detail, click here to see it.
This is a cone 6 transparent fritted glaze (converted from a Gerstley Borate one). Its B2O3 content is high, sourced by Ferro Frit 3134. Bubbles like this plague many potters, many just keep trying new glazes until one works, or give up on never finding one.
The first obvious problem is the frit, it starts melting at 1350°F, while plenty of gases are still being generated. Such a bubble-trapper is a non-starter in an industrial continuous fast-fire kiln. They need late melters. But potters have flexible firing, so what could be done? The firing could be slowed down, leading up to 1350. It could be held at top temp, then either slow cooled or a drop-and-hold.
And the recipe?
Notice the big bubbles; they started as little ones that merged. Given enough time, big ones break at the surface, but only under the right conditions: Low enough melt viscosity and surface tension. That's not happening. Strangely, old recipes sourcing high boron from Gerstley Borate had surprisingly few problems with bubbles! Why? GB was its own fining agent. And its boron entered the melt much later than this frit. Plus, the melt developed unevenly, creating localized channels and variable viscosity zones for easier bubble escape. The larger bubbles could better move laterally by combinations of surface tension, layer thickness and temperature gradients, and downward movement that created shear. A frit is missing all of that.
Let's assume this glaze melt has high surface tension. It pulls liquid inward around each bubble, stabilizing them round and making rupture more difficult. But, if surface tension drops, even slightly, bubbles deform more easily and adjacent ones merge. What could help? Industrial technicians have found that surprisingly small changes can really help with bubble release.
-A small SiO2 or Al2O3 addition can delay surface sealing or change viscosity timing.
-Sourcing the CaO from wollastonite can help bubble coalescence and reduce melt surface tension.
-Zinc oxide often changes surface behavior more than expected. As little as 2% can alter the viscosity curve, surface tension and melt interface properties, weakening bubble walls and improving near-surface rupture.
-Although MgO stiffens the melt somewhat, it also changes bubble wall elasticity; even small additions can help bubbles merge better.
As usual with solutions found here, methodical testing is needed to find the best answer.
Videos
Links
| Materials |
Ferro Frit 3134
A frit with 23% B2O3. The most common of frits used in pottery in North America. Around the world, other companies make frits of equivalent chemistry. |
| Materials |
Gillespie Borate
A Gerstley Borate substitute that became available during the early 2000s and is still available in 2023. |
| Materials |
Gerstley Borate
Gerstley Borate was a natural source of boron for ceramic glazes. It was plastic and melted clear at 1750F. Now we need to replace it. How? |
| URLs |
https://digitalfire.com/gb
GerstleyBorate.com - The best place for info on Gerstley Borate This site of the original gerstleyborate.com website, online from 2005-2025. This is the original content at a new address. |
Got a Question?
Buy me a coffee and we can talk

https://backup.digitalfire.com, All Rights Reserved
Privacy Policy

